Brain scans show psilocybin may physically rewire depression-linked brain networks
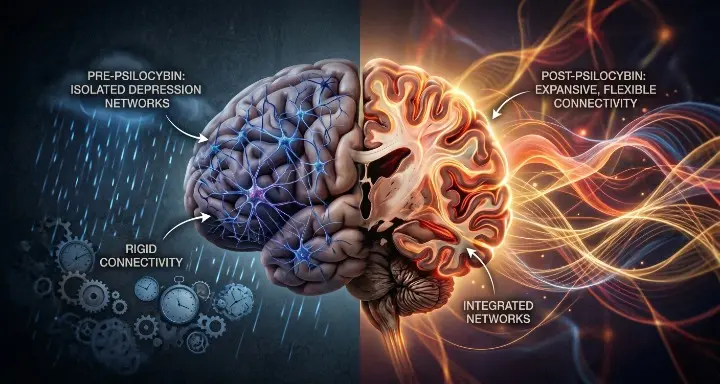
Your brain has a default setting, and for millions of people living with depression, PTSD, or addiction, that setting is stuck. A network of interconnected regions called the Default Mode Network keeps firing in rigid, looping patterns of self-criticism, rumination, and fear. Traditional antidepressants can dull the signal. What a new wave of neuroimaging research is revealing is that psilocybin and MDMA don’t just quiet the noise. They physically restructure the network itself.
This isn’t a metaphor for feeling better after a therapeutic session. It is a measurable, fMRI-confirmed reorganization of the brain’s architecture, documented weeks after the drug has left the body entirely.
What the Default Mode Network Actually Does
To understand why this matters, you need to understand what the Default Mode Network is and why it becomes a problem. The network anchors around four functional hubs: the medial prefrontal cortex, the posterior cingulate cortex, the precuneus, and the angular gyrus. When your brain has nothing specific to do, this network activates. It handles self-reflection, autobiographical memory, and mind-wandering.
In a healthy brain, this is useful. In a depressed brain, it becomes a trap. The network locks into overactive loops of negative self-referential thinking. In PTSD, it keeps the fear response perpetually online. In addiction, it generates cravings through memory-anchored reward associations. The problem isn’t the network itself. The problem is rigidity, a brain that cannot escape its own default patterns no matter how much the person consciously wants to.
What Psilocybin Does to the Locked Brain
The landmark neuroimaging evidence comes from a Nature Medicine study led by Robin Carhart-Harris, then at Imperial College London. The research assessed brain function in two separate clinical trials of depression, one in patients with treatment-resistant depression and one comparing psilocybin directly against escitalopram, one of the most widely prescribed antidepressants in the world.
The fMRI results were striking. In both trials, the antidepressant response to psilocybin was rapid and sustained, correlating directly with decreases in brain network modularity. In plain terms, the rigid, siloed structure of the depressed brain became more integrated and flexible. The higher-order networks rich in serotonin 2A receptors, the same receptors psilocybin binds to, became more interconnected. The brain was literally rewired.
The comparison with escitalopram made the distinction impossible to ignore. The antidepressant response to escitalopram was milder, and no changes in brain network organization were observed at all. The drug reduced symptoms through a different mechanism. Psilocybin changed the physical structure of how the brain communicates with itself.
The Reset Mechanism
Subsequent research has gone deeper into how this rewiring happens. When psilocybin is active in the brain, it triggers a suppression of hippocampal connectivity with the Default Mode Network. This persistent suppression appears to be a key mechanism behind both the pro-plasticity effects and the antidepressant outcomes. The brain is briefly freed from its own structural habits, and in that window of loosened connectivity, new pathways form.
Studies have also found that these changes correlate with the intensity of the altered state of consciousness experienced during the session. The more profound the experience, the more significant the network reorganization observed on scans days and weeks later. This is not a sedative effect or a numbing of the system. It is closer to a forced update of the operating system, one that the brain then maintains after the compound has cleared.
How MDMA Works Differently
MDMA does not suppress the Default Mode Network in the dramatic way psilocybin does. Its mechanism is distinct and in many ways complementary. Rather than restructuring the network itself, MDMA modulates the connectivity between emotion-regulating networks and the Default Mode Network’s regions, facilitating greater emotional openness and a measurable reduction in fear responses.
This is precisely why MDMA has shown such promise specifically for PTSD. The condition is fundamentally a disorder of fear memory. Traumatic experiences become encoded in a way that keeps the nervous system in a state of chronic threat. Standard therapy requires patients to revisit those memories directly, which often triggers overwhelming fear responses that shut down the therapeutic process before it can work.
MDMA changes the chemistry of that encounter. Under its influence, patients can access traumatic memories with reduced fear and greater emotional flexibility. The memory is retrieved, but the terror that normally accompanies retrieval is attenuated enough to allow genuine processing. The result, in phase 3 clinical trial data, is that 71% of veterans and first responders with PTSD showed lasting relief at the 12-month mark, a figure that dwarfs anything conventional pharmacotherapy has produced for the same population.
The Numbers That Changed the Conversation
The clinical outcomes running alongside the neuroimaging data are what have pushed this research from academic curiosity into a genuine regulatory conversation. A phase 3 randomized, double-blind, placebo-controlled trial published in Nature Medicine found that MDMA-assisted therapy produced significant reductions in PTSD severity scores compared to placebo with therapy, with 67% of participants no longer meeting the criteria for a PTSD diagnosis after treatment.
For psilocybin, the 12-month data on depression remission is sitting above 58%, with some trials reporting that a single high-dose session produces sustained improvements lasting longer than a year. For context, the remission rate for treatment-resistant depression using standard pharmacological approaches is typically below 30%, and those results rarely hold at 12 months without continued medication.
Why the FDA Story Is Complicated
The regulatory picture is where the science collides with institutional caution. The FDA rejected Lykos Therapeutics’ application for MDMA-assisted therapy in 2024, citing concerns about trial design and the difficulty of blinding participants in psychedelic studies, since the subjective experience of the drug makes it nearly impossible for participants not to know whether they received the active compound. This is a genuine methodological challenge unique to psychedelic research, not a reflection of the clinical data itself.
The rejection did not stop the field. More than three dozen psychedelics-related bills were introduced across over a dozen US states in the 2025 legislative session alone. Oregon and Colorado have both moved toward regulated therapeutic access frameworks. The science continues accumulating faster than the regulatory structures can process it.
What the Brain Scans Are Actually Telling Us
The deeper implication of this research goes beyond any specific condition. For decades, psychiatry has operated on the assumption that mental illness is primarily a chemical imbalance correctable by adjusting neurotransmitter levels over months of daily medication. The neuroimaging data coming out of psilocybin and MDMA research is telling a different story.
What these compounds appear to do is temporarily dissolve the structural rigidity that keeps a suffering brain trapped in its own patterns, and in doing so, create a window in which the brain can reorganize itself around healthier connectivity. The therapy conducted during and after that window is what shapes where the new connections form.
This is not pharmacology in the traditional sense. It is closer to neurological renovation, using a compound not as a daily maintenance drug but as a one-time catalyst for structural change. The fMRI data showing rewired networks weeks after a single session is not a side effect. It is the mechanism.
Sources:
1. Daws, R.E., Timmermann, C., Giribaldi, B., et al. Increased global integration in the brain after psilocybin therapy for depression. Nature Medicine, 2022; 28(4): 844–851. DOI: 10.1038/s41591-022-01744-z https://www.nature.com/articles/s41591-022-01744-z
2. Mitchell, J.M., Ot’alora G., M., van der Kolk, B., et al. MDMA-assisted therapy for moderate to severe PTSD: a randomized, placebo-controlled phase 3 trial. Nature Medicine, 2023; 29: 2473–2480. DOI: 10.1038/s41591-023-02565-4 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10579091/
3. Default Mode Network Modulation by Psychedelics: A Systematic Review. International Journal of Neuropsychopharmacology, 2022. DOI: 10.1093/ijnp/pyac074 https://pubmed.ncbi.nlm.nih.gov/36272145/
4. Carhart-Harris, R.L., et al. Psilocybin for treatment-resistant depression: fMRI-measured brain mechanisms. Scientific Reports, 2017; 7: 13187. DOI: 10.1038/s41598-017-13282-7 https://www.nature.com/articles/s41598-017-13282-7
5. Psychedelic Therapy in 2025: New Research Reveals Surprising Clinical Outcomes. GlobalRPH, October 2025. https://globalrph.com/2025/10/psychedelic-therapy-in-2025-new-research-reveals-surprising-clinical-outcomes/
6. Revealing Changes in Linear and Nonlinear Functional Connectivity After Psilocybin and Escitalopram Treatment in Patients with Depression. bioRxiv, March 2025. DOI: 10.1101/2025.03.05.641592 https://www.biorxiv.org/content/10.1101/2025.03.05.641592v1.full